Imagine you’re a paint manufacturer and one of your customers has complained that the product they received was defective. Your first step would be to investigate the complaint and identify the root cause of the problem. Your investigation could involve interviews with the client and employees and an in-depth analysis of any relevant data. In addition, you’d probably want to look at the defective batch closely to understand what was wrong with it and how it differed from the other batches you produce and the product specification. This thorough examination helps pinpoint exactly where the deviation occurred, whether during the formulation, production, or packaging stages.
Once you’ve identified the root cause of the problem, you can put a preventive action in place to prevent it from happening again. In this case, you might change your recipe or how your paint products are inspected before they’re shipped to customers. For example, implementing stricter quality checks or recalibrating machinery might be necessary to ensure consistent product standards. You might also update your training program to make sure all employees are aware of new quality control measures, which ensures they understand the importance of adherence to procedures. Furthermore, enhancing communication protocols may also be required to guarantee timely reporting and response to any potential issues. The Corrective and Preventive Action (CAPA) process briefly illustrated above is vital to quality management. Why and how does CAPA fit into a quality management system – we’re about to find out.
Why should manufacturers use CAPA?
While the above example is about fixing problems when they arise, CAPA is also about analyzing information, and improving processes and procedures to limit error reoccurrence. By identifying trends and taking proactive steps to address them, you can improve your overall manufacturing process and prevent future issues. The CAPA process is instrumental in driving continuous improvement across production facilities, optimizing both efficiency and consistency.
Manufacturers often find that by implementing a comprehensive CAPA system, greater transparency emerges within their operational workflows, allowing for better monitoring and evaluation of each phase of production. In addition to resolving existing problems, CAPA lays the foundation for a culture of quality and accountability within manufacturing environments.
By embedding CAPA into everyday practices, businesses can significantly diminish the probability of faults, leading to higher product standards and more satisfied customers. This is especially crucial in highly regulated industries where compliance and product quality are paramount and non-conformances can lead to significant financial and reputational damage. By tackling issues at their root, organizations can implement tailored measures to ensure sustainable improvements, thereby bolstering their competitive advantage in the marketplace.
The CAPA process can help organizations to:
- Prevent errors from happening again
- Improve product quality and safety
- Reduce costs associated with rework, scrap, and customer complaints
- Increase customer satisfaction
Furthermore, it can address any type of problem, whether customer complaints, product defects, process issues, audit findings, or anything else that is below par.
A successful CAPA process
For CAPA to be effective, it must be well-designed and consistently followed. There are four essential elements of a successful CAPA system:
- Identifying problems: This is usually done through product inspection, complaint handling, audits, or other monitoring activities.
- Root cause analysis: Once a problem has been identified, it’s essential to understand the root cause. This can be done through various methods such as brainstorming, process mapping, 5 Why Analysis, or Ishikawa Diagrams.
- Corrective actions: Once the root cause has been identified, corrective actions can be taken to address the issue. These actions should be designed to prevent the problem from happening again.
- Preventive actions: In addition to corrective actions, preventive actions can be taken to address potential problems before they occur. This might involve process improvements, design changes, or training employees on new procedures.
Another step to consider is monitoring and improvement. CAPA systems should be regularly tracked for effectiveness, and refinements should be implemented to improve further where necessary.
Main steps in every CAPA process
There are typically several different steps involved in the CAPA process. While each of these steps can be further expanded on, in general, the first stage is always information gathering.
CAPA Information Gathering
CAPA is typically started when a problem or non-conformance is identified. Gathering information may involve conducting interviews, reviewing data or records, or observing process steps. It could also include data analysis to identify trends. This data may help to identify the root cause of the problem and further actions that could be taken to prevent it from happening again.
The goal of CAPA information gathering is to ensure that all relevant information about the issue is collected so that appropriate next steps can be taken. To achieve this, it is crucial to employ a structured approach that includes meticulous documentation of every detail related to the issue, which may involve leveraging advanced tracking systems to capture and organize data efficiently.
In certain contexts, stakeholders and subject matter experts might be engaged in this phase to provide insights and validate findings. Additionally, conducting a thorough review of historical records can unveil patterns that may have been overlooked during daily operations. Such comprehensive analysis not only aids in tackling the current issue but also enhances the organization’s ability to anticipate and mitigate similar problems in the future, contributing to a robust quality management system.
CA vs. PA
The information-gathering phase identifies details of the issue, its impact, and the underlying root cause. The CA or corrective action phase is next and is focused on removing the root cause so that the situation is stabilized, and services are restored. Sometimes, the CA phase can be about fixing the incident with a workaround. Think for example about shipping a new product, while there have been no structural improvements yet.
During the PA (preventative action) phase, actions are taken to address any similar future instances proactively. PA includes a defined change control process where the potential impact is measured. During a change meeting, decisions are made on whether to proceed or wait based on the overall business and client impact. It is important to note that early prevention can help mitigate the need for correction entirely.
Let’s illustrate these last two phases with an example. Think of a manufacturer that ships a product with the wrong label. In this case, the CA would be to collect the products with the wrong label and relabel them. The PA would be to add an inspection point in the process to double-check whether the right labels are fed into the filling machine.
How CAPA fits into Quality Management
CAPA is part of a more extensive quality management system known as PDCA (plan-do-check-act) or the Deming Cycle. PDCA is a continuous improvement cycle that helps businesses identify problems, correct them, and prevent their recurrence.
CAPA is the “check” and “act” part of PDCA. After you’ve identified a problem and put a plan in place to correct it, CAPA helps you check to see if the program is working and make any necessary adjustments. It also enables you to take preventive action to keep the problem from happening again.
When done correctly, CAPA can be an extremely effective tool for improving quality and preventing problems. However, it’s important to note that CAPA is only as good as the data that’s used to identify problems. For CAPA to be effective, businesses must have a robust system in place for collecting data and identifying trends.
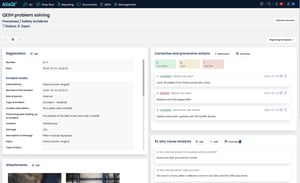
In AlisQI, CAPA is integrated into the bigger picture of QA/QESH management and is not a stand-alone activity or tool. Manufacturers start corrective and preventive actions on the QESH problem-solving page. This standardized dashboard, based on the 8-D methodology can easily be tailored and provides all the components they need to collect data, analyze, follow up and report incidents.
Effectiveness of CAPA
By addressing problems at their root cause, CAPA can prevent minor issues from snowballing into major ones. Additionally, by taking proactive steps to prevent future problems, CAPA can help organizations avoid disruptions and maintain high levels of quality.
Implementing a CAPA system can also be beneficial from a compliance perspective. Many quality standards, such as ISO 9001 and AS9100, require organizations to have a CAPA system in place. Additionally, many regulatory bodies, such as the FDA, expect organizations to have a CAPA system to address quality issues.
However, having a CAPA program in place doesn’t guarantee its effectiveness. The CAPA process must be continuously tracked to ensure it works as intended.
Evaluate the effectiveness of individual actions. Did the corrective actions indeed take away the customer’s problem? What was the lead time? Did the preventive action indeed take away the root cause?
When these actions deliver in a timely fashion, you have a well-oiled CAPA machine. This will be the heart of your continuously improving organization.
Final Thoughts
There are many benefits of implementing a CAPA system in your business. By addressing problems and preventing them from happening again, you can improve your product quality, reduce customer complaints, and avoid costly recalls.
The quickest way to familiarize yourself with CAPA and the AlisQI problem-solving page is by means of the interactive tour.